
NEWS
Professional design, development and production of digital encoder, electronic switch and high and low voltage connector
Information classification >
More value same price——TTC Gold Pink Keyswitch
2022-03-07 17:23

Two years ago today, on March 7, 2020, TTC released the Gold Pink Keyswitch, the first in the industry to adopt long spring structure, realizing fresh linear keyswitch feel with soft touch and strong rebound. Using TTC patented double side wall dustproof structure keyswitch core(patent number: CN201620103314.9), which makes it more stable, dustproof and reduce the possibility of shaking. Once listed, it has been loved and sought after by the majority of users, and has been rated as "the best keyswitch of the year" and "annual five-star excellent product" by many media such as hot technology, IN peripherals, peripherals, etc.


Two years later , on March 7, 2022, TTC gold pink keyswitch is upgraded. Under the condition of unchanged structure and parameters, the same "dripping with gold and silver" process as TTC honey keyswitch is adopted, that is, the silver plating process of anti-oxidation coating is added on the outer surface of the terminal to improve the oxidation resistance and durability of keyswitch.
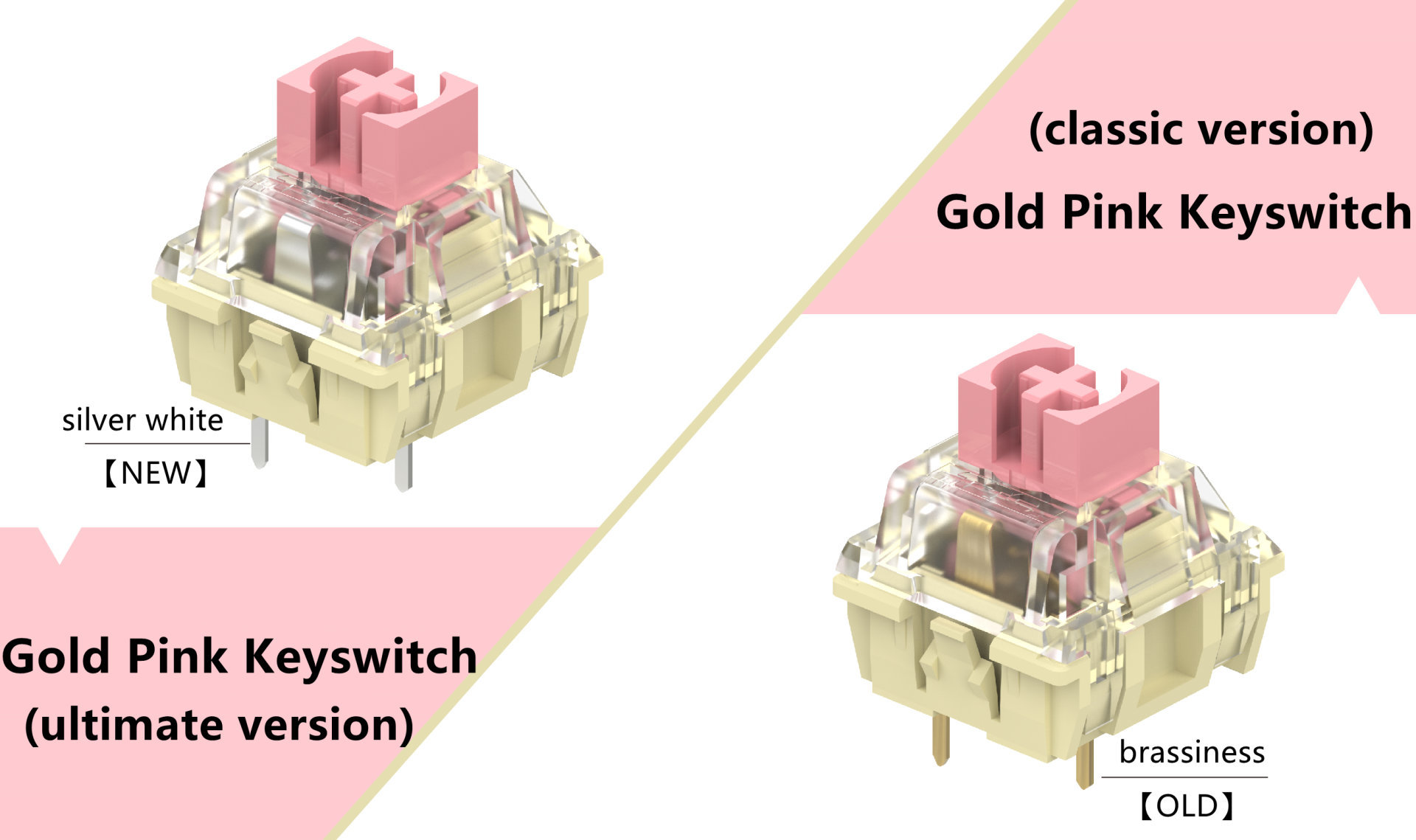
As we all know, the materials of the two terminal pins of the keyswitch are made of copper alloy, and the phenomenon of copper oxidation and blackening is inevitable. With the wide application of hot plug, the number of hot plug is increasing, and the time uncertainty of keyswitch exposed in complex environment is also increasing, resulting in more and more serious oxidation and blackening of exposed terminal pins. TTC gold pink keyswitch is the first to be upgraded. The silver plating process of anti-oxidation coating is added on the outer surface of the terminal, which has obvious anti-oxidation effect and no price increase.

The tips of keyswitch
First, why is the surface of copper easily oxidized, discolored and blackened?
Why is the surface of copper workpiece (copper alloy) always very easy to change color and blacken, and even serious corrosion phenomena such as verdigris?

First of all, let's know about copper from its atomic structure:
Copper is a transition element, its chemical symbol is Cu, English name is copper, atomic number is 29.
The schematic diagram of copper atomic structure is as follows:

The electron layer of copper atom: K-L-M-N; Its electron layer distribution: 2-8-18-1; Electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 3D¹⁰ 4s¹
As we all know, the number of outermost electrons is an important basis to determine the chemical properties of elements!
Copper is more likely to lose an electron and be oxidized. It also leads to the copper parts in the absence of protective measures under the condition of easy oxidation discoloration.
Chemical properties and reactions of copper:
1. Reaction with oxygen: copper is a less active heavy metal. It does not combine with oxygen in dry air at room temperature. When heated, it can produce black copper oxide:

If it continues to burn at very high temperature, it will produce red Cu2O:

2. Reaction with air (reaction with O2, H2O and CO2): after being exposed to humid air for a long time, a layer of copper green (basic copper carbonate) will be slowly formed on the copper surface. Copper Green can prevent further corrosion of metal, and its composition is variable.
Copper is oxidized in humid air:

Copper is oxidized and discolored in humid air:

The above chemical reactions are the basic principle of oxidation discoloration of copper parts in the actual production and processing process. Copper parts are prone to oxidation and discoloration under corrosive conditions such as humidity, so they need to be protected.
How to prevent copper oxidation discoloration?
Copper protection type: physical protection, chemical passivation, chemical coating protection, etc. Physical protection mainly includes spraying antirust paint, sealing wax and so on; Chemical passivation protection mainly includes traditional chromate passivation (hexavalent chromium has strong carcinogenicity and has been banned), organic corrosion inhibitor passivation (representative product is mebes ms0423, which does not contain any toxic heavy metals); Chemical coating protection mainly includes tin plating, nickel plating, chromium plating, zinc plating and so on. The main purpose of these protective measures is to separate the corrosive medium from the copper substrate and prevent further corrosion.
Second, why the keyswitch terminal surface silver plating can improve the oxidation resistance and durability of keyswitch?
The physical and chemical properties of silver are relatively stable, with good thermal and electrical conductivity. In principle, silver and oxygen cannot have direct chemical reaction at room temperature, but silver can oxidize and blacken slowly due to long-term contact with hydrogen sulfide in the air. Chemical equation: 4Ag +2H2S + O2= 2Ag2S + 2H2O (the principle of silver metamorphism and blackening at room temperature).
The concentration of hydrogen sulfide in normal air is very low, because the monitoring of hydrogen sulfide concentration in air is one of the important indicators to evaluate air pollution. The maximum allowable concentration of harmful substances in the air of workshops in China (tj36-79) is 10mg / m ³. The maximum allowable concentration of harmful substances in the atmosphere of residential areas in China (tj36-79) is 0.0110mg/m ³ (primary value).
Therefore, adding anti-oxidation coating and silver plating process on the outer surface of the terminal can effectively improve the anti-oxidation and durability of the keyswitch.
(The above description comes from the network)
Related news






